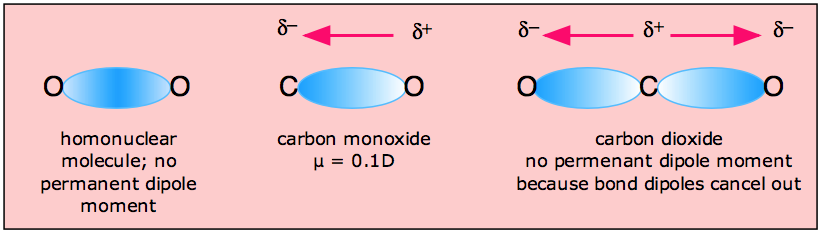
 It is also found in particle physics in the measurement of possible dipole moments of electrons and neutrons for example. A quantitative measurement of the bonds polarity or molecules polarity is termed as dipole moment. Usually used for atoms and molecules, the notion of dipole moment is used to describe the electric fields of such systems as well as Van der Waals forces. Its existence is strongly related tothe symmetry properties of a molecule. If the two centers do notcoincide the molecule has a permanent dipole moment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
